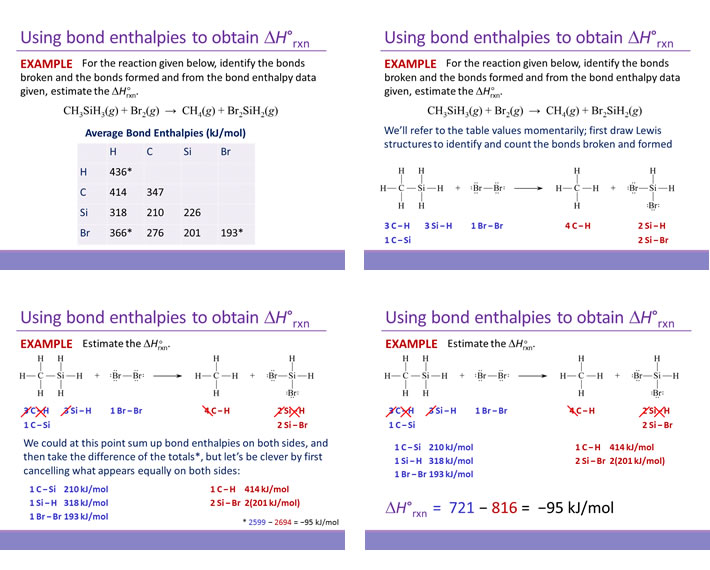
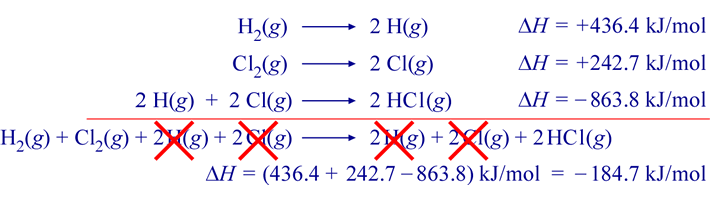SOLVED: Check Your Understanding: Use the following thermochemical equations to find the heat of formation of diamond: C(diamond) + O2(g) -> CO2(g) ΔH° = -39544 kJ 2 CO(g) + O2(g) -> 2

Calculate the standard enthalpy of the reactions 2C (graphite) + 3 H2(g)→ C2H6(g) from the following Δ H^∘ value (i) C2 H6(g) + 72 O2 (g)→ 2 CO2(g) + 3 H2 O (