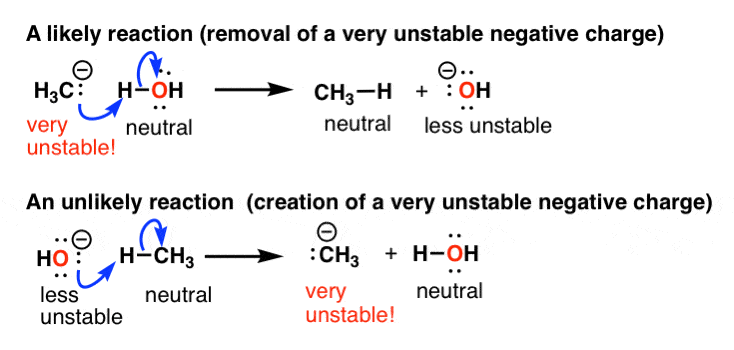
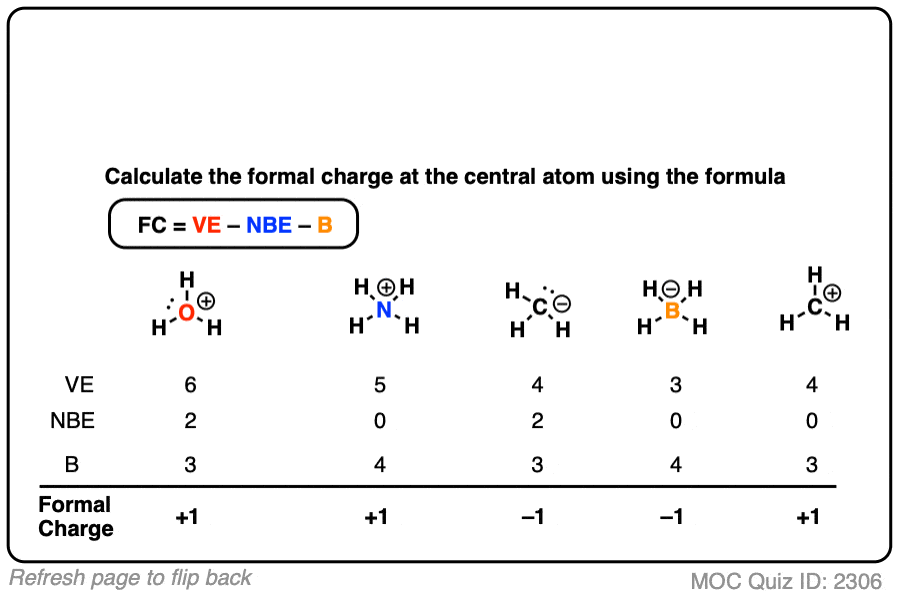
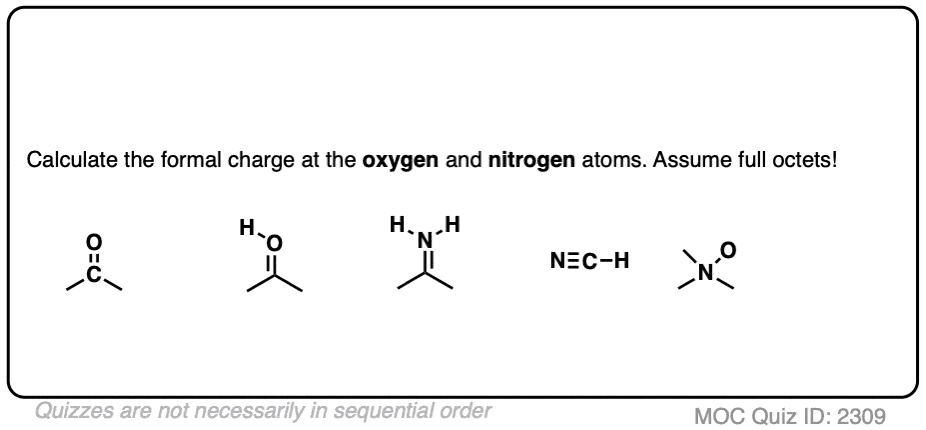
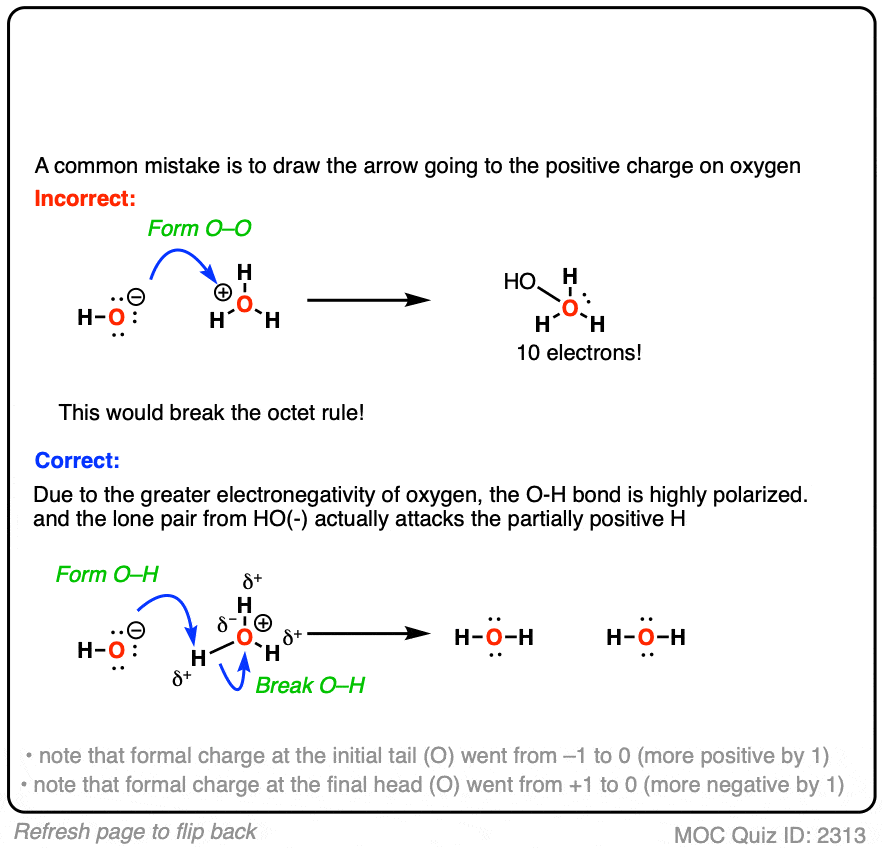
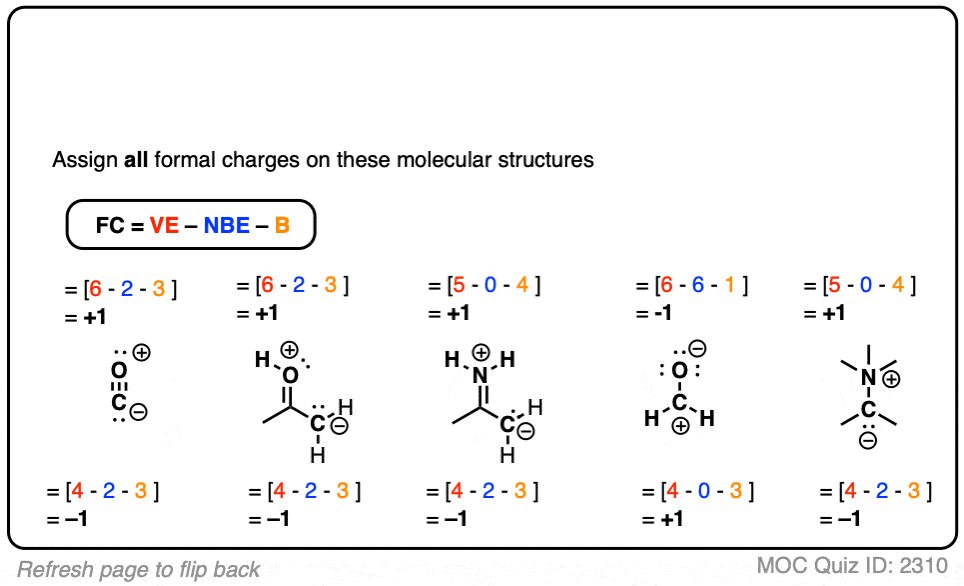
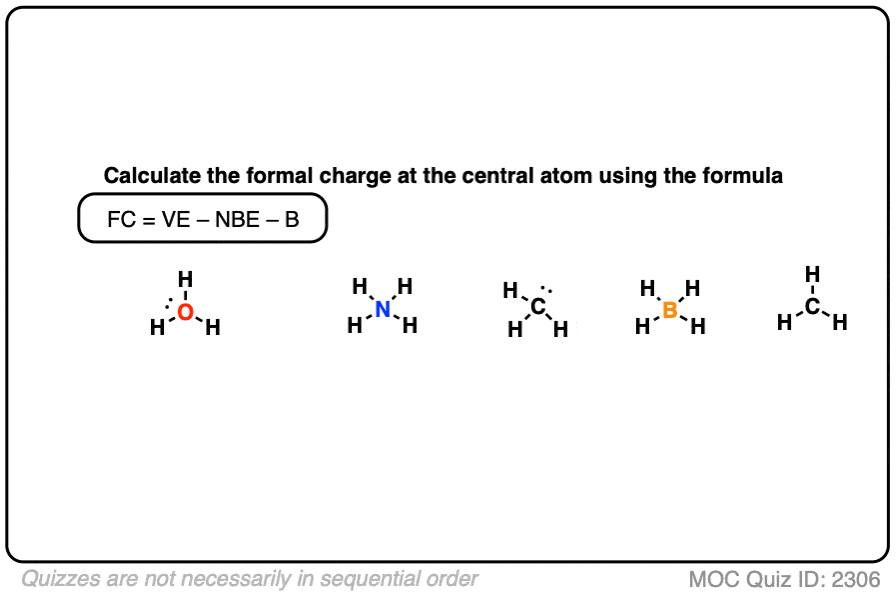
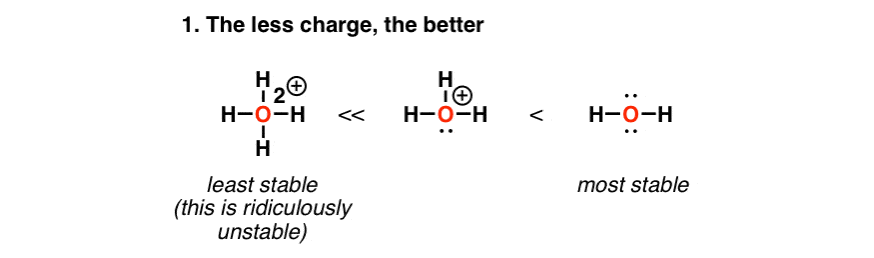
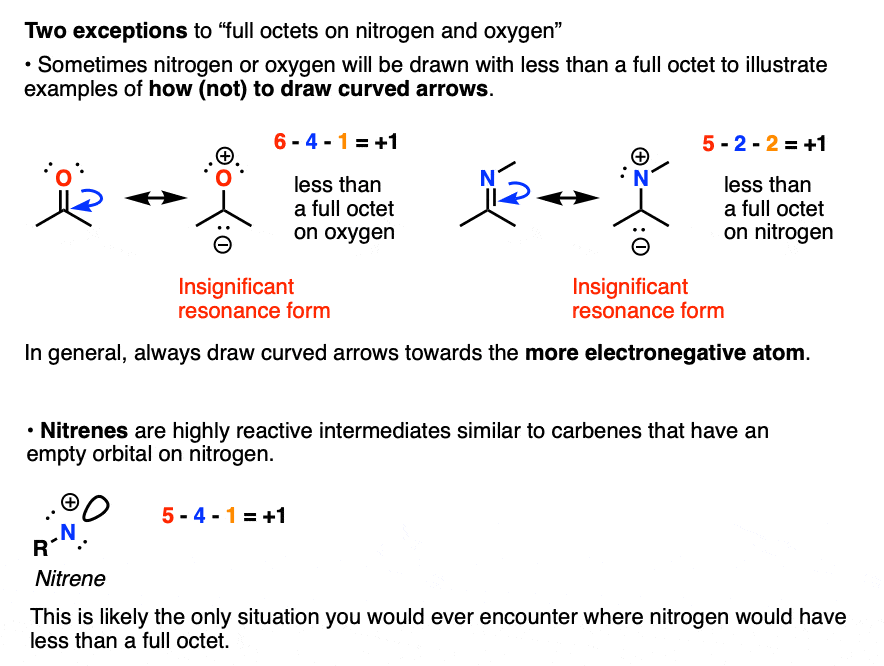
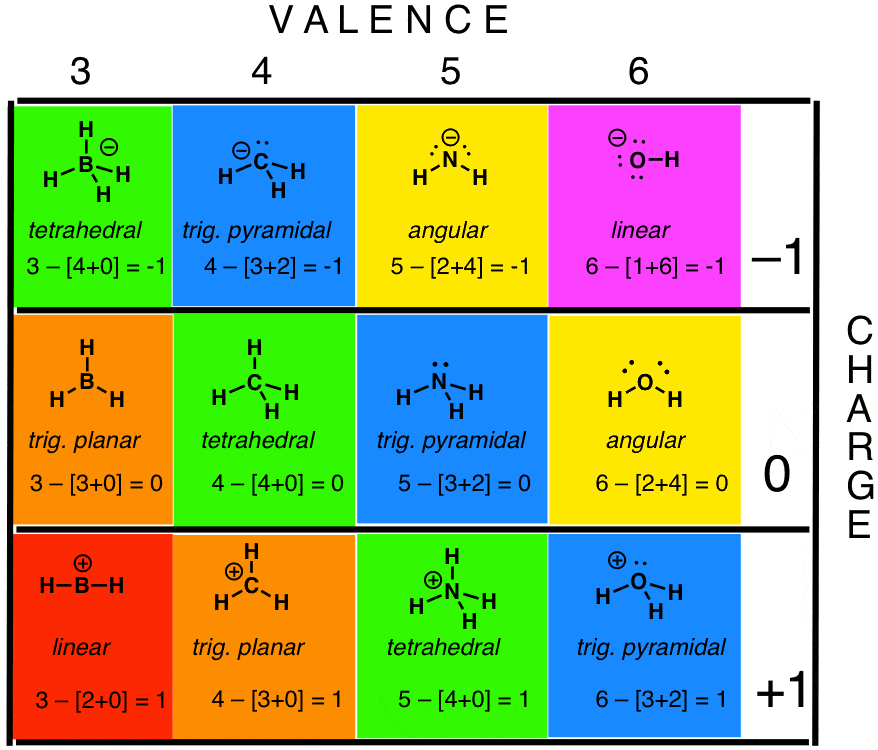
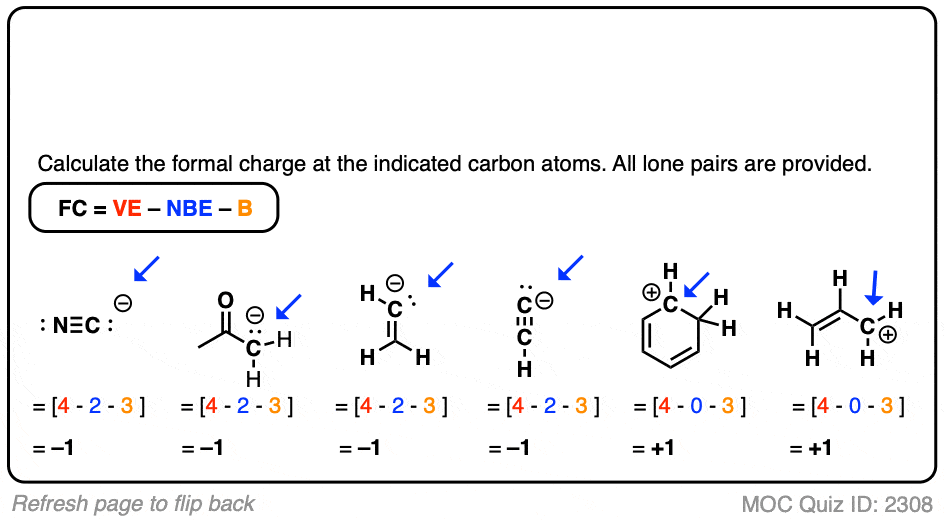
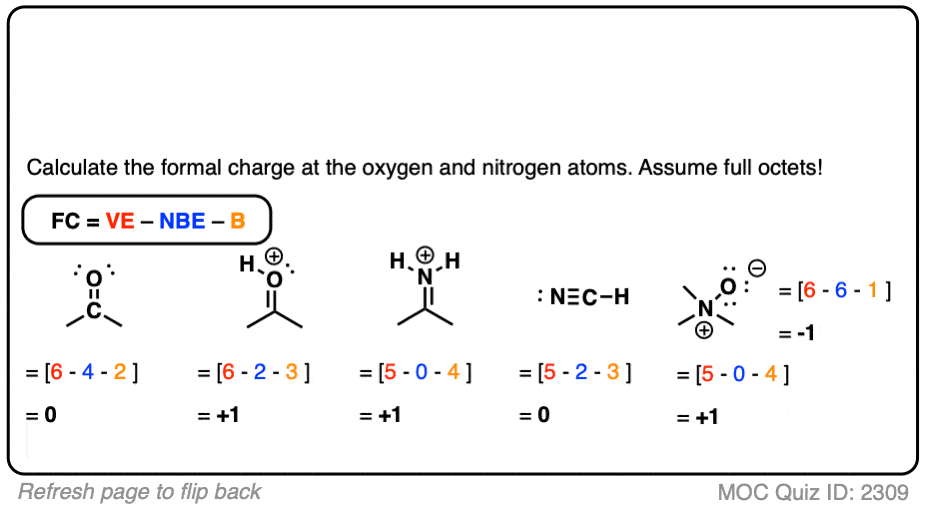
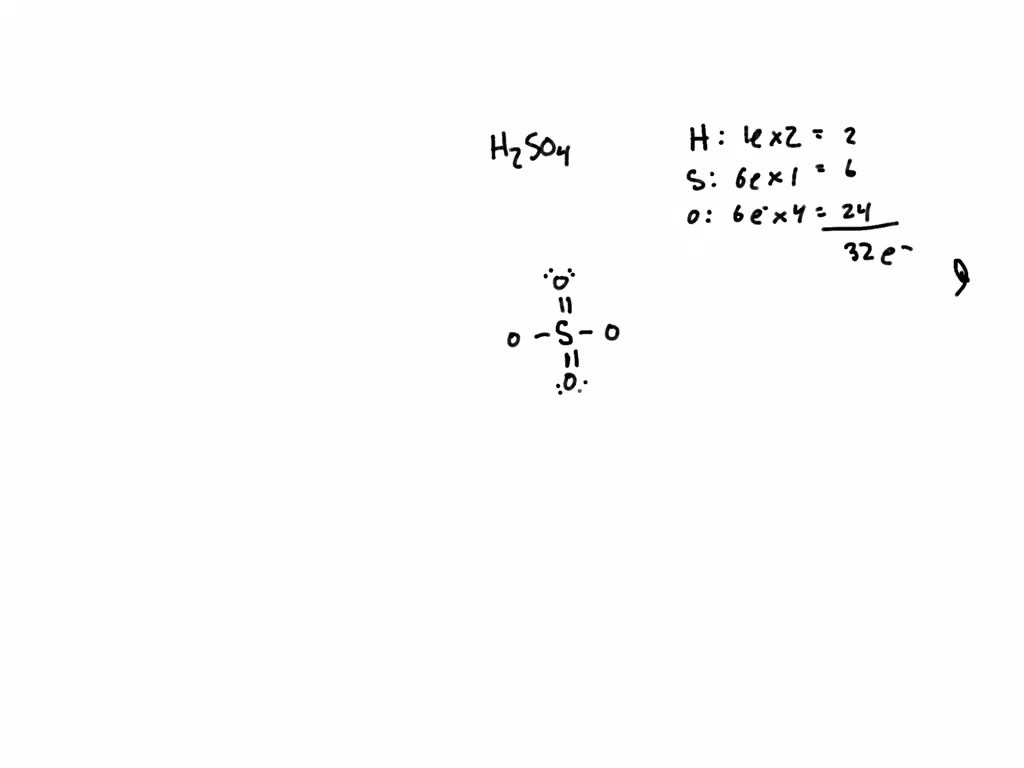
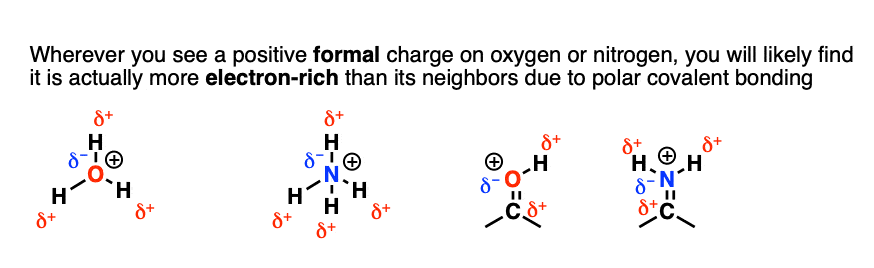Self-consistent calculations of charge self-trapping energies: A comparative study of polaron formation and migration in PbTiO3 | Computational Materials Group @ Chalmers

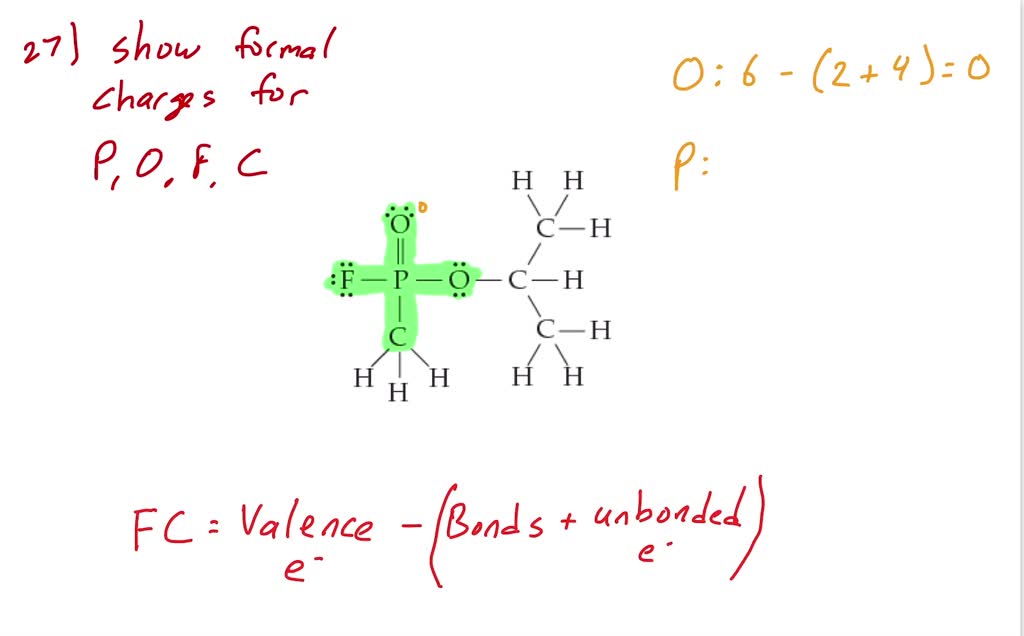
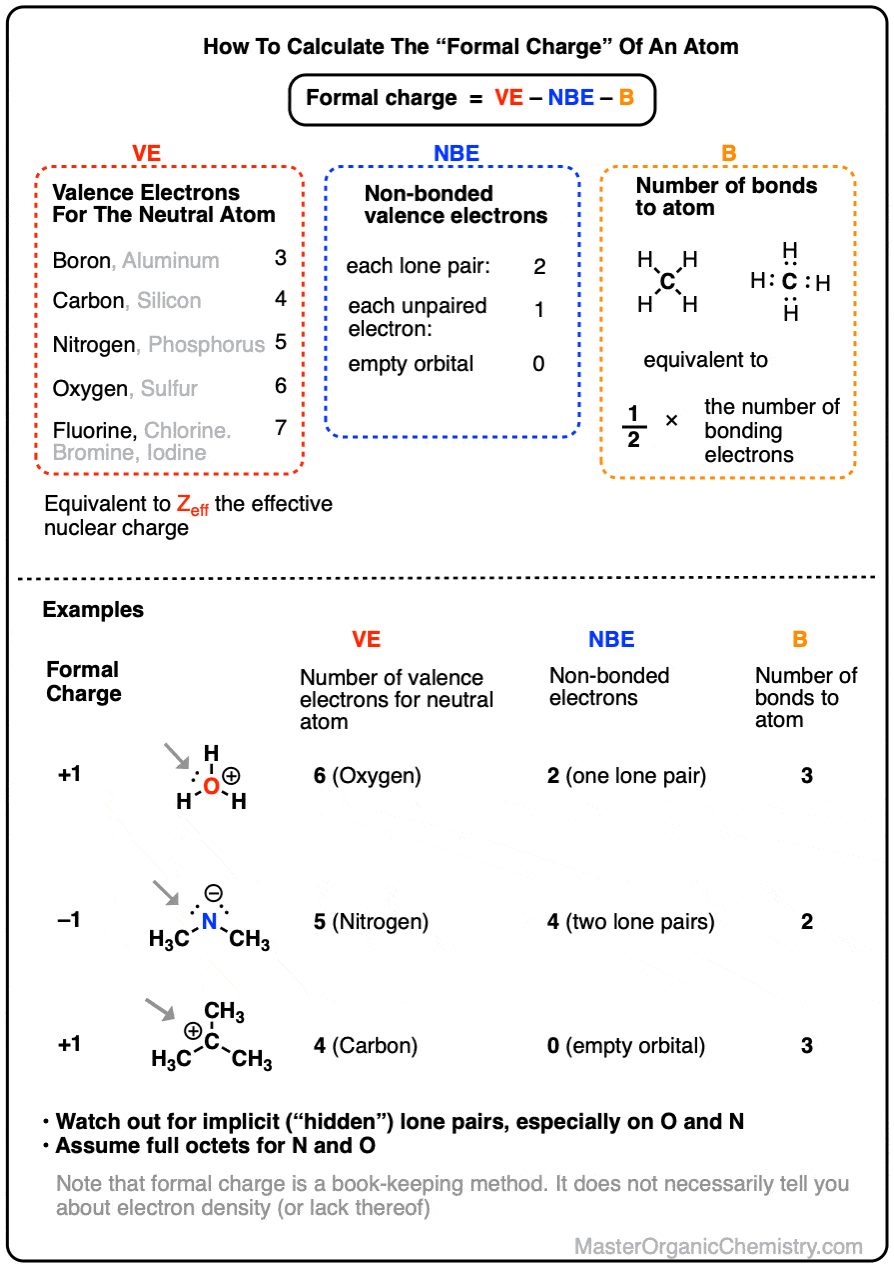
SOLVED: What are the formal charges on atoms B, and D respectively in the following structure? A= D=0 A=0,B=- ,D =-1 A=+1,B=-1,D =-1 A=0,B= 0,D =-1
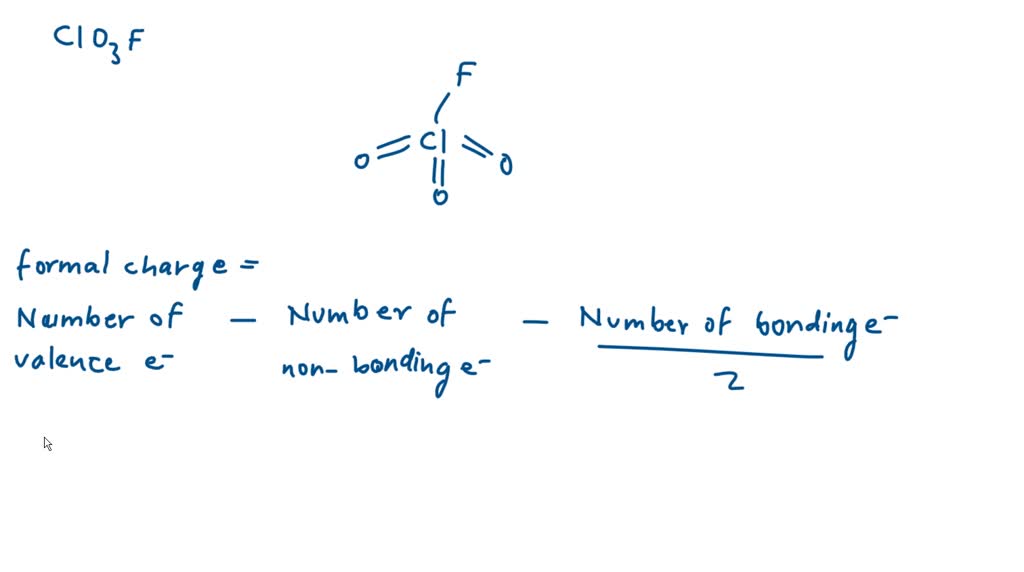
SOLVED: calculate the formal charge of Chlorine (Cl) in the following compound. Show yourcalculation!ClO3F
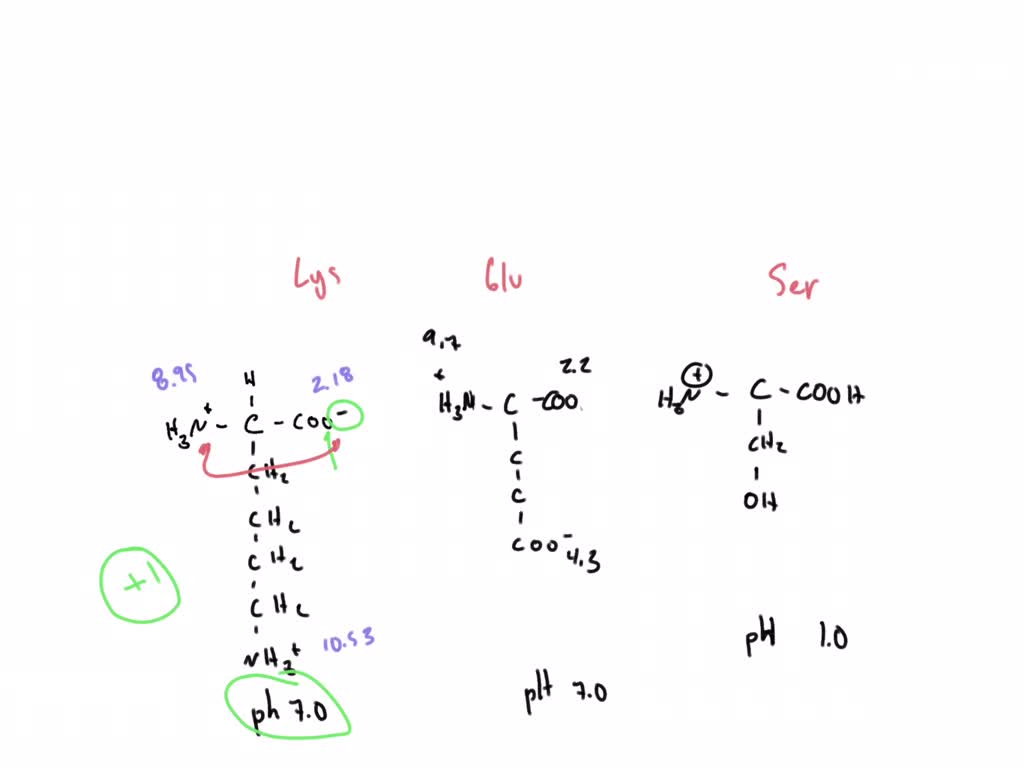
SOLVED: 'Calculate the charge and draw the structure of the predominant ion for: Lysine at pH 7.0 CH,CH,CH-CH,NHz) (ii) Glutamic acid at pH 7.0 (R= CHCH-COOH) (iid) Serine at pH 1.0 (R=- -