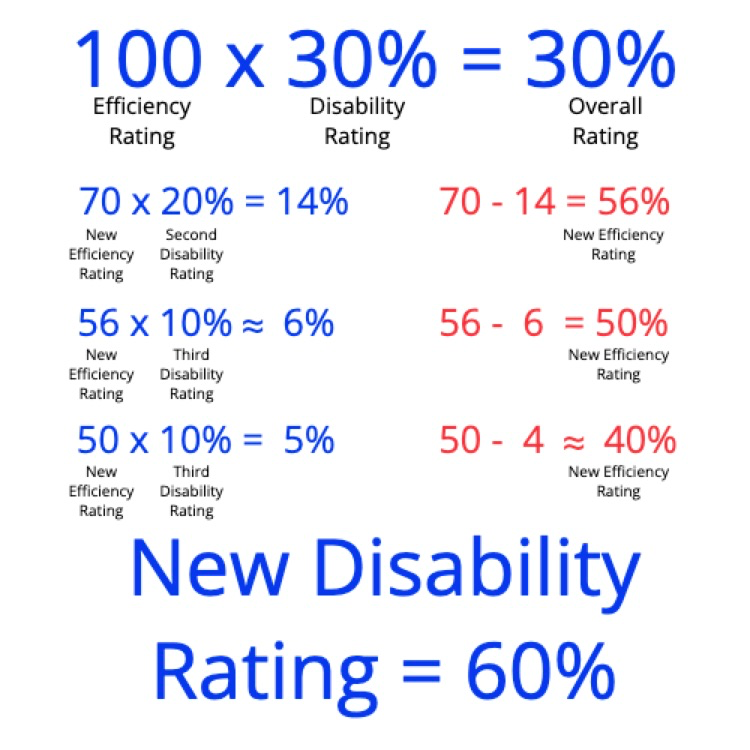
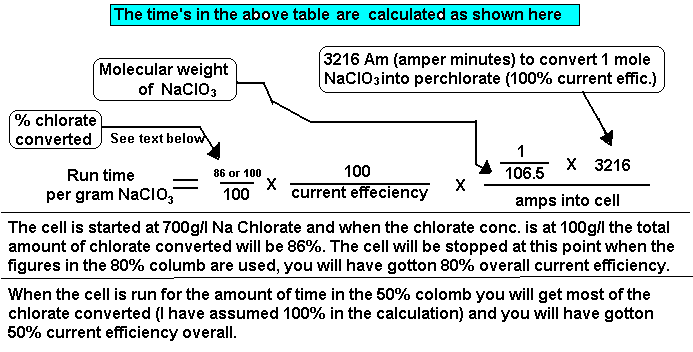
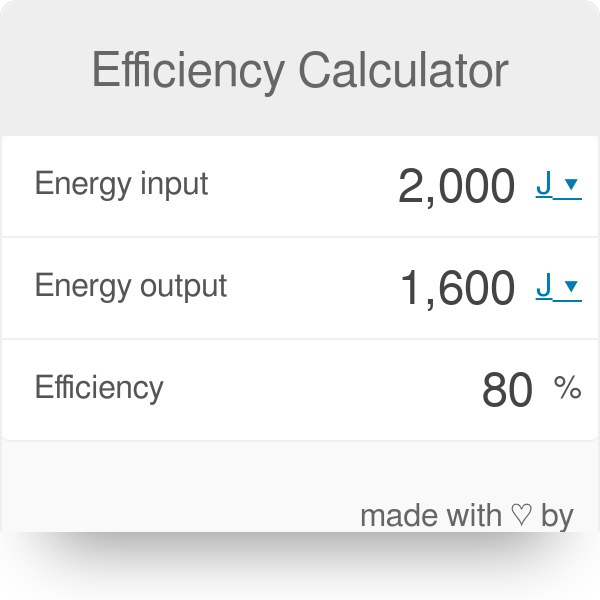
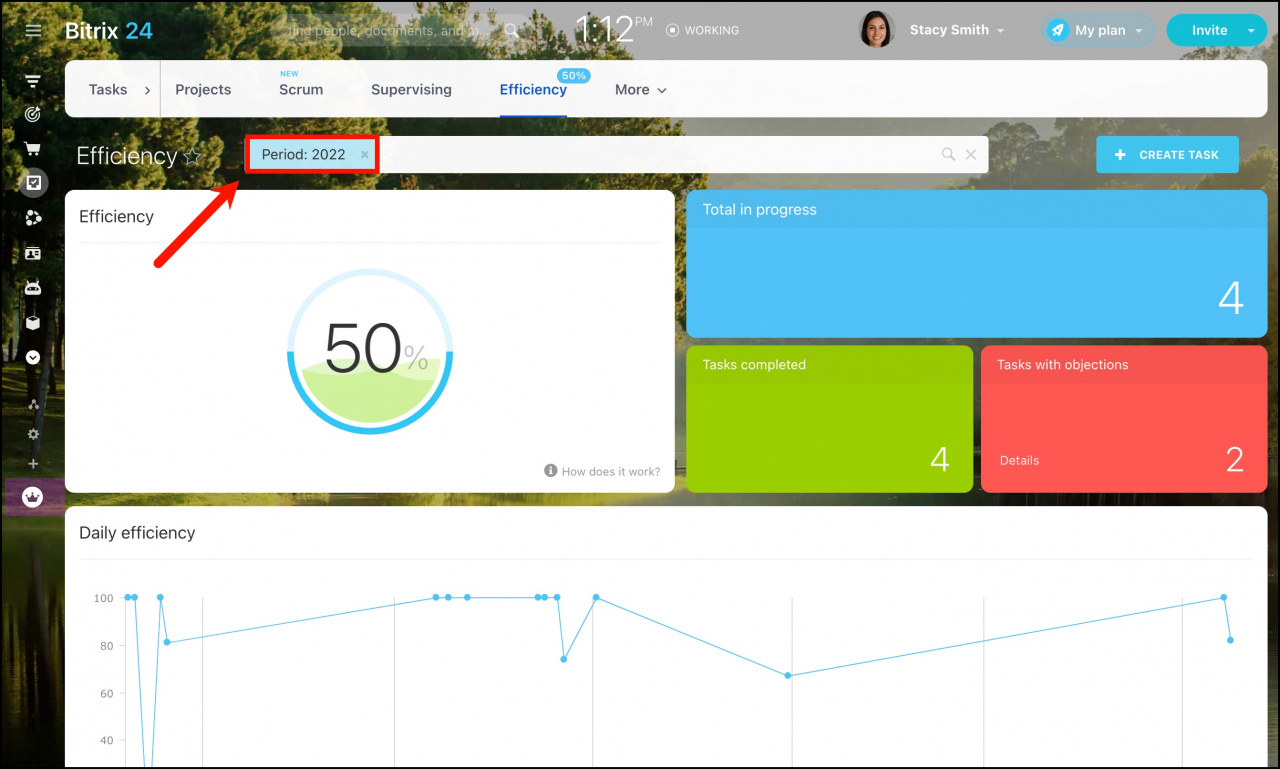
SOLVED: Calculate the heat absorbed by the water; using the formula in the introduction of this experiment: For water; C = 4.18 Jlg"C. Change your final answer to kJ Calculate the heat

SOLVED: Reaction Mass Efficiency third way calculate the effici ncy of reaction takes into ccount both percent yield and atom nomy: This method of evaluating the efficiency of reaction is called Reaction
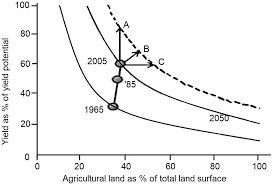


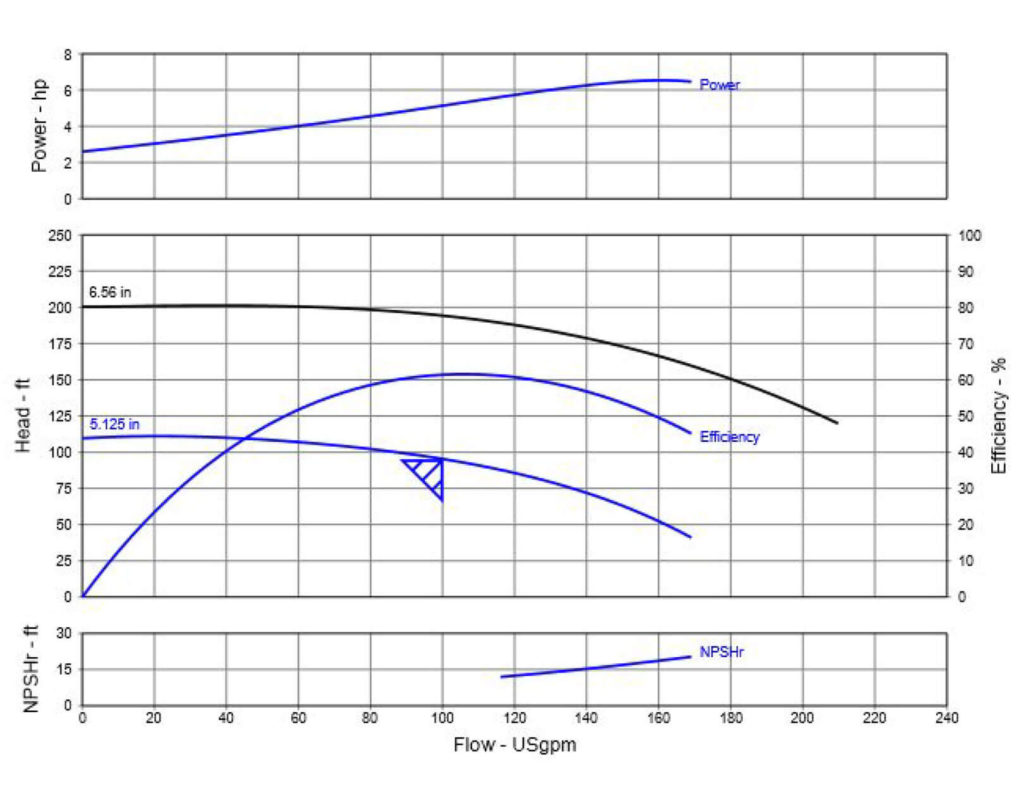

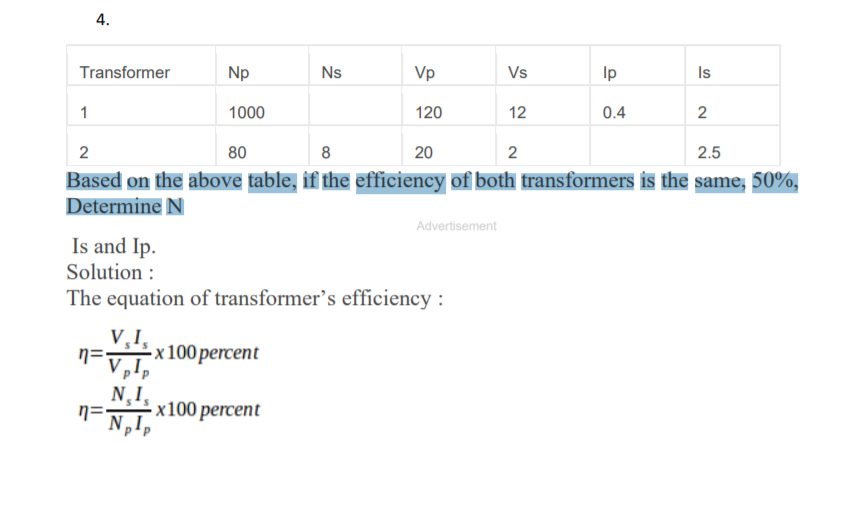
:max_bytes(150000):strip_icc()/Roi-5c4a640a34204da285b4ef1c98970be0.png)

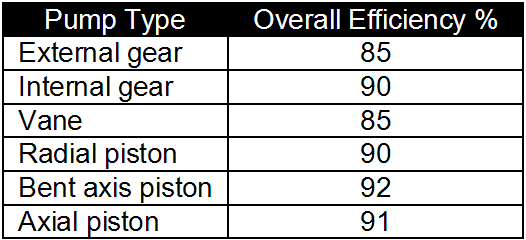
:max_bytes(150000):strip_icc()/Investopedia_Returnoninvestmentformula_colorv1-6d281839c5814e109e316ebbbb61a5bd.png)