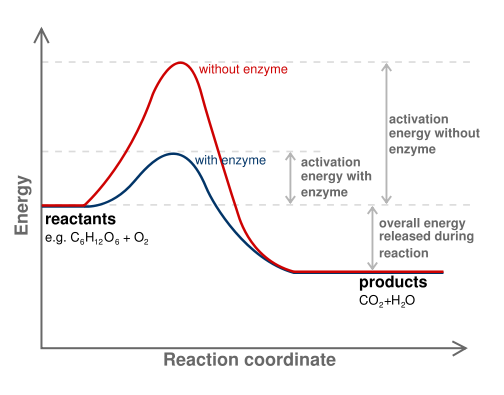
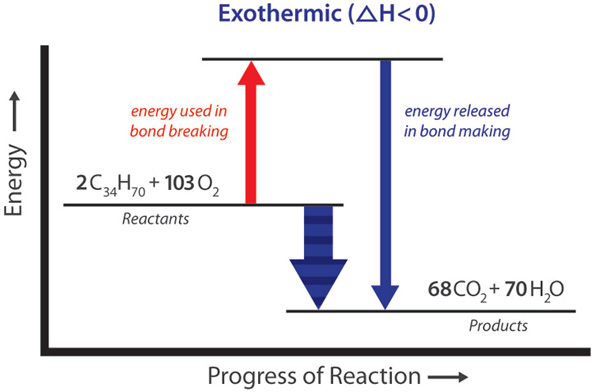
Review of the Decomposition of Ammonia to Generate Hydrogen | Industrial & Engineering Chemistry Research

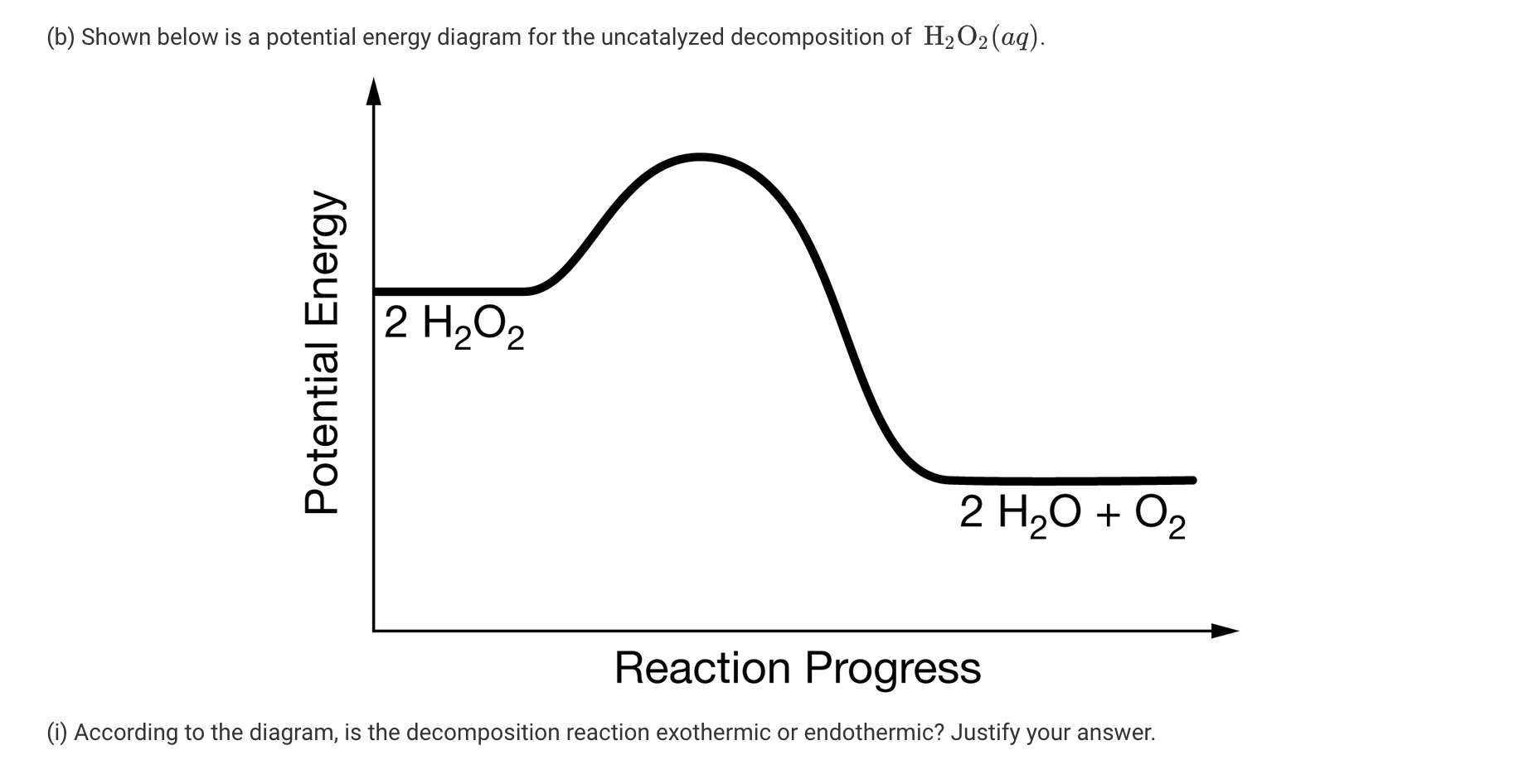
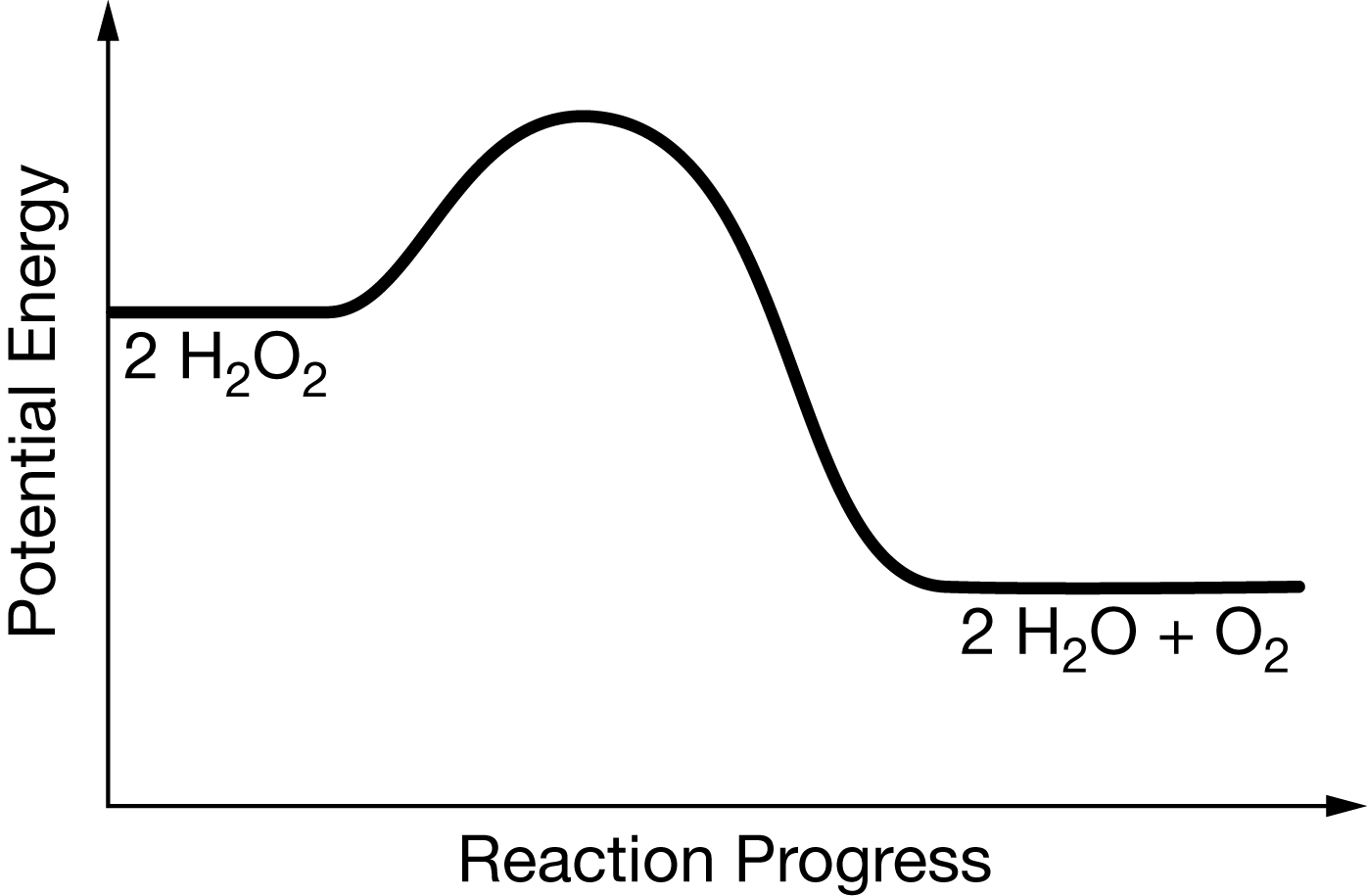
SOLVED: If 2 mol of hydrogen peroxide is decomposed, the reaction enthalpy (ΔH) is -54.04 kJ. If 0.814 g of H2O2 decomposes, how much heat (kJ) will be produced? Is this reaction

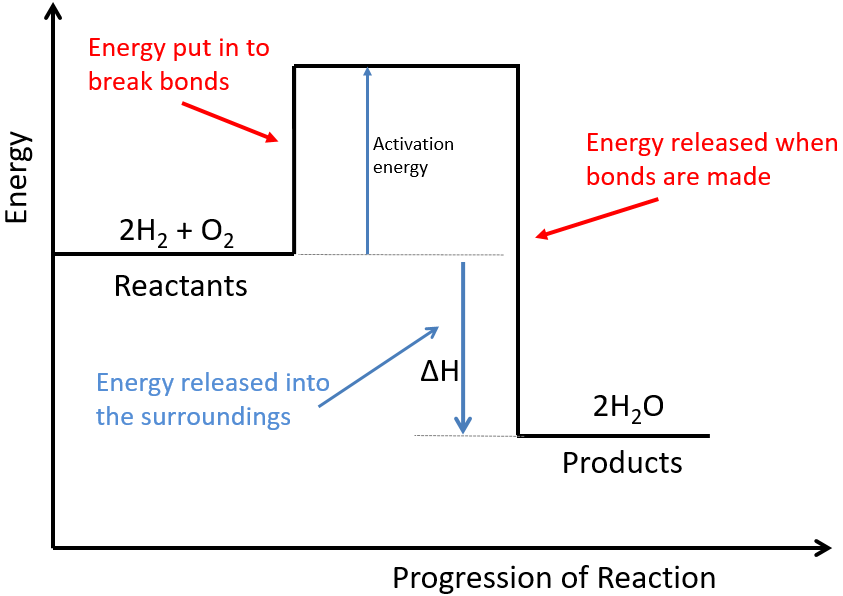
Hydrogen peroxide decomposes into water and oxygen by the following reaction: 2 H 2 O 2 ( a q ) 2 H 2 O ( l ) + O 2 ( g ) ,

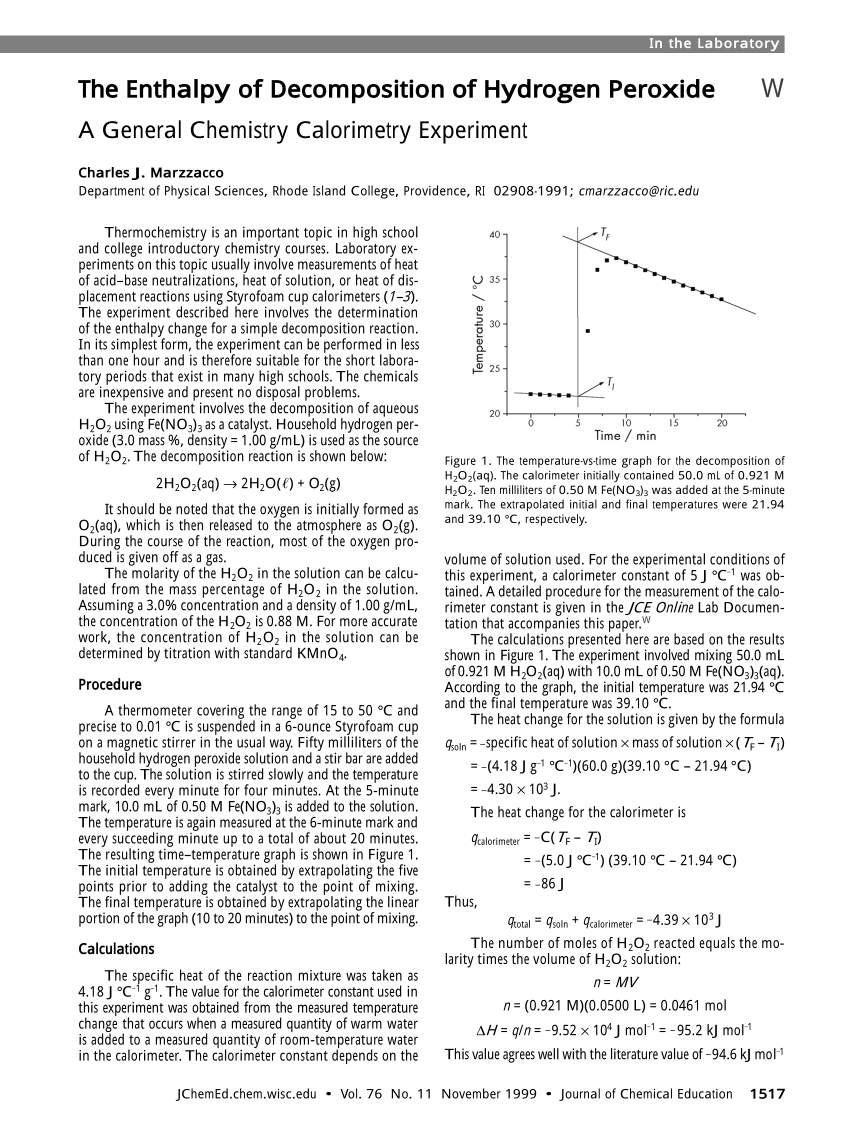
The temperature-vs-time graph for the decomposition of H 2 O 2 (aq).... | Download Scientific Diagram