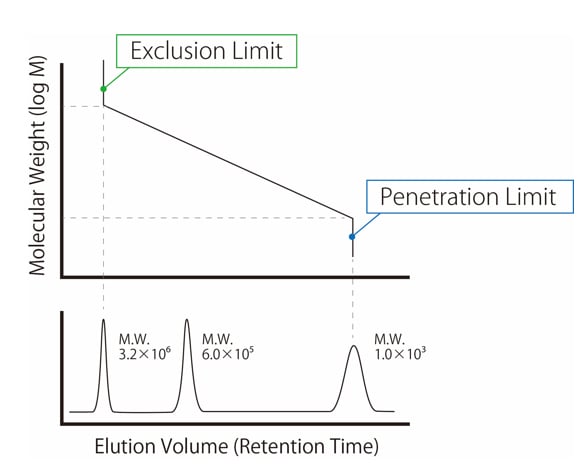
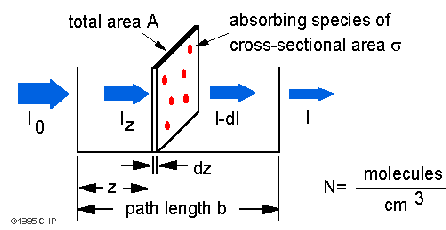
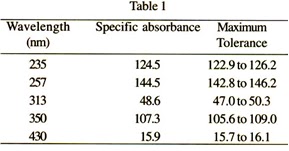
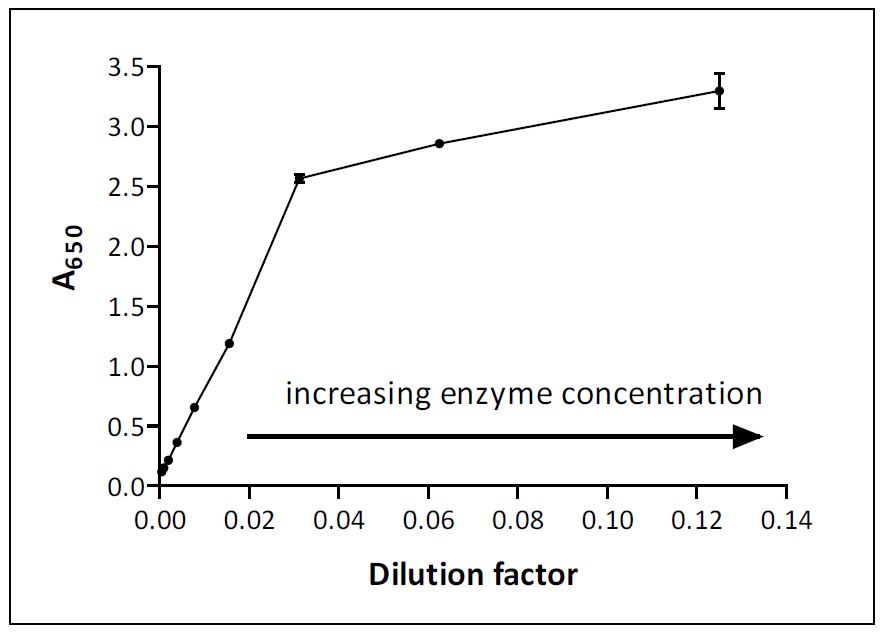
Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions

SOLVED: A spectrophotometric assay was t0 bz canied out on compound with concentration 5.04 ppm and molar absorptivity 9.60 10' L mol ! cm - Calculate the percent transmittance if the measurement

SOLVED: Use Beer's law to convert the given absorbance to molar concentration, then to percent concentration. e = 97000 M-1cm-1 Molecular weight = 792.8 grams/mol To calculate percent error, you will compare

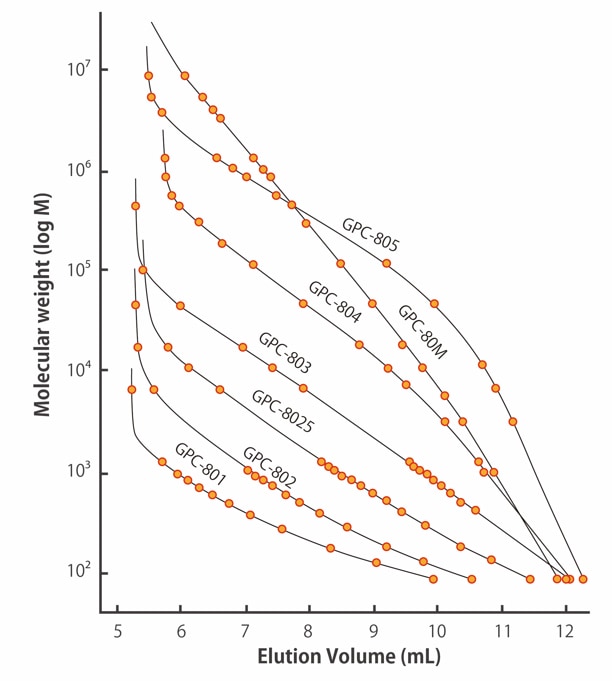
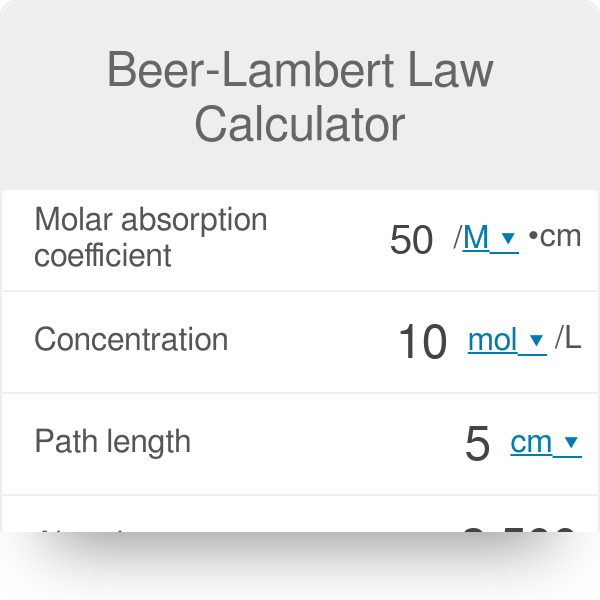
![Solved [8 marks] The concentration of chemical solutions can | Chegg.com Solved [8 marks] The concentration of chemical solutions can | Chegg.com](https://media.cheggcdn.com/media/964/96422413-45fd-4286-ae08-f3bb9f8cebd7/php5wTrL4)